Advertisement |
 |
Synthesis design tool that facilitates innovation
ICSYNTH from InfoChem, a Nature Research company, is a powerful computer-aided synthesis design software that can help you find the best route to synthesis. The advanced chemoinformatics algorithm can perform retrosynthetic analysis on target molecules and generate many possible synthetic routes, even if the target molecules are novel and/or unpublished.
FIND OUT MORE |  | |
 |
 |
TABLE OF CONTENTS
|
April 2017 Volume 9, Issue 4 |
 |  |  |
 |  Editorial Editorial
 News and Views News and Views
 Articles Articles
 In Your Element In Your Element |  | Advertisement |  |  |  |
Focus on membrane materials for separations
Membrane materials offer many practical advantages for purification and separation applications. Nature Materials presents a Focus which highlights the most promising new membrane materials that offer competitive performance capabilities, and discusses how to transfer such materials and processes to industry.
Read the Focus | |
|
|
 |
|
 |
 |
| Advertisement |
 |
ELECTRON MICROSCOPY FOR MATERIALS - THE NEXT TEN YEARS
Presented by: Zhejiang University | Nature | Nature Materials
The conference will feature sessions on nanomaterials, functional materials, structural material, soft matter and biomaterials, and techniques development.
May 27-29, 2016 | Zhejiang, China
Register now! |  | |
 |
| |
Editorial |  Top Top |
 |
 |
 |
Life, but not as we know it p297
doi:10.1038/nchem.2763
There are many unanswered questions regarding how the biomolecules and biomechanical processes that define life came to be. A collection of Articles in this issue show how intermediates in RNA synthesis might have formed and how the initiation and evolution of RNA replication might have occurred. |
 |
News and Views |  Top Top |
 |
 |
 |
|
 |
| Advertisement |
 |
- Reach a potential audience of 10.8 million users* via nature.com
- Be seen first by candidates — your position will appear at the top of related searches for 60 days and be displayed prominently on the Naturejobs home page as Job of the Week.
- Plus appear as 'Featured' in search results
| | |
 |
| |
Articles |  Top Top |
 |
 |
 |
A prebiotically plausible synthesis of pyrimidine β-ribonucleosides and their phosphate derivatives involving photoanomerization pp303 - 309
Jianfeng Xu, Maria Tsanakopoulou, Christopher J. Magnani, Rafal Szabla, Judit E. Šponer et al.
doi:10.1038/nchem.2664
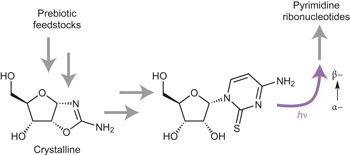
Although ribose aminooxazoline has been shown to be a potential intermediate in prebiotic pyrimidine ribonucleotide synthesis, a route by which this could occur has remained elusive. Now, a remarkably efficient photoanomerization reaction has been investigated by theory and experiment. The new route affords enantiomerically pure ribonucleotides when the starting material is enantioenriched.
Chemical compounds |
 |
 |
 |
Prebiotic synthesis of phosphoenol pyruvate by α-phosphorylation-controlled triose glycolysis pp310 - 317
Adam J. Coggins and Matthew W. Powner
doi:10.1038/nchem.2624
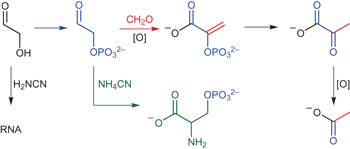
Chemical reconstitution of the triose glycolysis pathway is controlled by α-phosphorylation and provides a generational link between prebiotic ribonucleotide synthesis, triose glycolysis and serine metabolism. Now, research suggests that unification of nucleotide synthesis and triose metabolism may have been a fundamentally important step towards the origins of life.
Chemical compounds |
 |
 |
 |
A viscous solvent enables information transfer from gene-length nucleic acids in a model prebiotic replication cycle pp318 - 324
Christine He, Isaac Gállego, Brandon Laughlin, Martha A. Grover and Nicholas V. Hud
doi:10.1038/nchem.2628
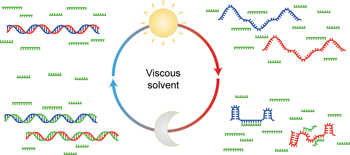
An unanswered question in the RNA world scenario is how sequence information could be transferred during replication of duplex RNA. Without the aid of sophisticated enzymes, strand reannealing occurs more quickly than template-directed synthesis. Now, a plausible prebiotic solution to this problem is presented, in which a viscous solvent enables information transfer from a gene-length double-stranded template. |
 |
 |
 |
Simple peptides derived from the ribosomal core potentiate RNA polymerase ribozyme function pp325 - 332
Shunsuke Tagami, James Attwater and Philipp Holliger
doi:10.1038/nchem.2739
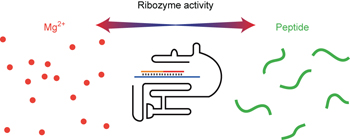
Lysine-rich peptides from the ribosomal core and derived homolysine decapeptides of either L-, D- or mixed chirality have now been shown to enhance RNA polymerase ribozyme activity at low magnesium concentrations, accelerate ribozyme evolution and enable templated RNA synthesis within membranous protocells. |
 |
 |
 |
Atomically precise organomimetic cluster nanomolecules assembled via perfluoroaryl-thiol SNAr chemistry pp333 - 340
Elaine A. Qian, Alex I. Wixtrom, Jonathan C. Axtell, Azin Saebi, Dahee Jung et al.
doi:10.1038/nchem.2686
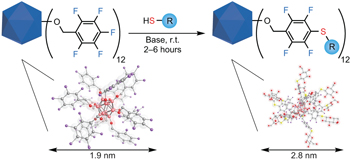
The operational simplicity of modifying the surfaces of thiol-capped gold nanoparticles has been a hallmark of their success in materials chemistry, despite having limited control over the surface composition. Now, SNAr chemistry on activated perfluoroaromatics has been shown to mimic this simplicity and allow for the synthesis of atomically precise nanomolecules.
Chemical compounds
See also: News and Views by Majewski et al. |
 |
 |
 |
Dynamics of the triplet-pair state reveals the likely coexistence of coherent and incoherent singlet fission in crystalline hexacene pp341 - 346
Nicholas R. Monahan, Dezheng Sun, Hiroyuki Tamura, Kristopher W. Williams, Bolei Xu et al.
doi:10.1038/nchem.2665
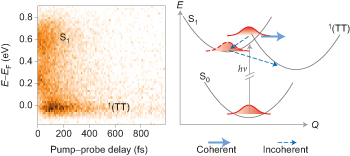
Singlet fission — the splitting of a singlet exciton into two triplets — is a process that could be exploited to improve the power conversion efficiency of solar cells. Spectroscopic data now suggest that coherent and incoherent mechanisms for singlet fission in crystalline hexacene coexist and occur on different timescales. |
 |
 |
 |
Surfactant-free single-layer graphene in water pp347 - 352
George Bepete, Eric Anglaret, Luca Ortolani, Vittorio Morandi, Kai Huang et al.
doi:10.1038/nchem.2669
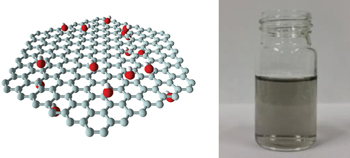
Aggregation usually prevents dissolution of graphene in water. Now, hydroxide ion adsorption has been shown to allow the stabilization of true single-layer graphene in water — with no surfactant required — so long as the liquid is degassed beforehand. The resulting aqueous dispersions can contain high concentrations of exfoliated graphene that are stable for several months. |
 |
 |
 |
Computational design of self-assembling cyclic protein homo-oligomers pp353 - 360
Jorge A. Fallas, George Ueda, William Sheffler, Vanessa Nguyen, Dan E. McNamara et al.
doi:10.1038/nchem.2673
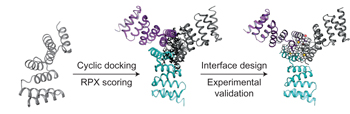
A computational method to design cyclic protein homo-oligomers has been developed. Using this approach, a series of idealized repeat proteins incorporating designed interfaces that direct their assembly into complexes possessing cyclic symmetry were fabricated. 15 out of 96 oligomers that were characterized experimentally were shown to be consistent with the computational model. |
 |
 |
 |
Remote C−H alkylation and C−C bond cleavage enabled by an in situ generated palladacycle pp361 - 368
Juntao Ye, Zhihao Shi, Theresa Sperger, Yoshifumi Yasukawa, Cian Kingston et al.
doi:10.1038/nchem.2631
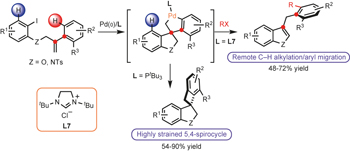
Existing methods for C–H activation depend on pre-installed directing groups, the removal of which poses a practical limitation on the use of these reactions in synthesis. Now, a remote-selective C−H alkylation reaction of arenes using an in situ generated spiropalladacycle has been shown to furnish benzofurans and indoles without the need for a directing group.
Chemical compounds
See also: News and Views by Murakami & Ishida |
 |
 |
 |
Real-time molecular scale observation of crystal formation pp369 - 373
Roy E. Schreiber, Lothar Houben, Sharon G. Wolf, Gregory Leitus, Zhong-Ling Lang et al.
doi:10.1038/nchem.2675

Homogeneous crystal nucleation has now been observed by transmission electron microscopy in real time on a molecular scale. Countercation-dependent observations of polyoxometalate proto-crystal formation confirm existence of a higher energy classical molecular attachment mechanism, as well as a lower energy two-step mechanism via an intermediate dense phase. |
 |
 |
 |
Aldehydes as alkyl carbanion equivalents for additions to carbonyl compounds pp374 - 378
Haining Wang, Xi-Jie Dai and Chao-Jun Li
doi:10.1038/nchem.2677
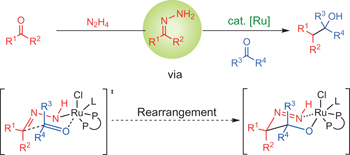
Methods utilizing renewable feedstocks are critical to accessing molecules of industrial importance in light of the present ecological and economic climate. Here, it is shown that umpolung reactivity of carbonyl compounds can be used for nucleophilic additions to yield a diverse array of valuable alcohols as an alternative to using stoichiometric organometallic reagents.
Chemical compounds |
 |
 |
 |
Entomopathogenic bacteria use multiple mechanisms for bioactive peptide library design pp379 - 386
Xiaofeng Cai, Sarah Nowak, Frank Wesche, Iris Bischoff, Marcel Kaiser et al.
doi:10.1038/nchem.2671
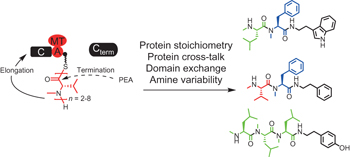
Nature has evolved a variety of different mechanisms to generate chemical diversity; however, the reactions responsible for generating such diverse chemical libraries are often not clear. Now, the mechanisms employed by entomopathogenic bacteria for the biosynthesis of a large family of bioactive peptides have been identified. These include substrate promiscuity, enzyme cross-talk and enzyme stoichiometry.
Chemical compounds |
 |
 |
 |
Seven enzymes create extraordinary molecular complexity in an uncultivated bacterium pp387 - 395
Michael F. Freeman, Maximilian J. Helf, Agneya Bhushan, Brandon I. Morinaka and Jörn Piel
doi:10.1038/nchem.2666
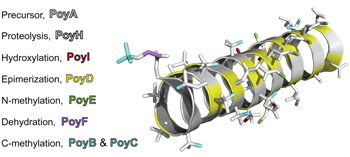
Polytheonamides are giant peptide toxins produced by the uncultivated sponge bacterium Entotheonella factor. The biosynthesis of polytheonamides involves up to 50 post-translational modifications. Now, heterologous expression in Escherichia coli and Rhizobium hosts have shown that a minimalistic, iterative enzyme set introduces this exceptional molecular complexity via epimerizations, C-/N-methylations, hydroxylations, dehydration and proteolytic maturation. |
 |
 |
 |
An iron-catalysed C–C bond-forming spirocyclization cascade providing sustainable access to new 3D heterocyclic frameworks pp396 - 401
Kirsty Adams, Anthony K. Ball, James Birkett, Lee Brown, Ben Chappell et al.
doi:10.1038/nchem.2670
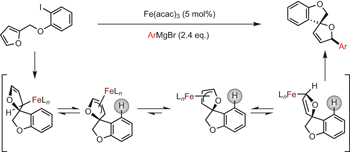
The use of earth-abundant metals in catalysis is a prerequisite for the development of sustainable synthetic chemical processes for the future. Now, biomass-derived chemicals can be used in an efficient iron-catalysed reaction to make complex spirocycles. The resulting products contain two asymmetric centres that are furnished in high stereoselectivity.
Chemical compounds |
 |
In Your Element |  Top Top |
 |
 |
 |
Iterations of ytterbium p402
Alasdair Skelton and Brett F. Thornton
doi:10.1038/nchem.2755
Alasdair Skelton and Brett F. Thornton examine the twisting path through the several discoveries of ytterbium, from the eighteenth century to the present. |
 |
 Top Top |
 |
 |
 |  |  |  |  |  | Natureevents is a fully searchable, multi-disciplinary database designed to maximise exposure for events organisers. The contents of the Natureevents Directory are now live. The digital version is available here.
Find the latest scientific conferences, courses, meetings and symposia on natureevents.com. For event advertising opportunities across the Nature Publishing Group portfolio please contact natureevents@nature.com |  |  |  |  |  |
|
 |


No comments:
Post a Comment