Advertisement |
 |
Nature Reviews Chemistry is now live!
Nature Reviews Chemistry provides both an introduction to chemists embarking on a new topic of investigation and thought-provoking, in-depth sections for the expert. The first issue is now published and is free to view online for a limited time.
Read the first issue |  | | |
 |
 |
TABLE OF CONTENTS
|
February 2017 Volume 13, Issue 2 |
 |  |  |
 |  Obituary Obituary
 Research Highlights Research Highlights
 News and Views News and Views
 Perspective Perspective
 Brief Communications Brief Communications
 Articles Articles
|  | Advertisement |  |  |  | 
Unexplored opportunities in the druggable human genome
This poster presents a categorization of human proteins based on the amount of data on them, highlighting a knowledge deficit and indicating novel drug discovery opportunities.
Download the Poster free online
Produced with support from:
Illuminating the Druggable Genome Knowledge Management Center
(IDG KMC) | | |
|
|
 |
|
 |
 |
Obituary |  Top Top |
 |
 |
 |
Susan Lindquist (1949-2016) p127
Tricia R Serio
doi:10.1038/nchembio.2305
Susan Lindquist passed away on 27 October 2016, far too early for those who marveled at her brilliance through her many contributions to science, for all who knew her directly, and especially for her former trainees.
|
 |
Research Highlights |  Top Top |
 |
 |
 |
RNA modification: Reading Sex-lethal | Biosynthesis: Terrifically tailored peptides | Pancreatic development: Changing identity | RNA structure: Untying Zika's knots
|
News and Views |  Top Top |
 |
 |
 |
| |
 |
Perspective |  Top Top |
 |
 |
 |
The pharmacological regulation of cellular mitophagy pp136 - 146
Nikolaos D Georgakopoulos, Geoff Wells and Michelangelo Campanella
doi:10.1038/nchembio.2287
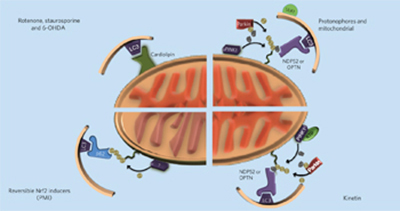
This perspective discusses recent progress in the development of pharmacological tools that initiate mitophagy and spare mitochondrial function and focuses on promising approaches to identify improved reagents.
|
|
|
 |
Brief Communications |  Top Top |
 |
 |
 |
|
 |
 |
 |
|
 |
Articles |  Top Top |
 |
 |
 |
|
 |
 |
 |
|
 |
 |
 |
|
 |
 |
 |
|
 |
 |
 |
|
 |
 |
 |
|
 |
 |
 |
|
 |
 |
 |
|
 |
 |
 |
Inhibition of innate immune cytosolic surveillance by an M. tuberculosis phosphodiesterase pp210 - 217
Ruchi Jain Dey, Bappaditya Dey, Yue Zheng, Laurene S Cheung, Jie Zhou et al.
doi:10.1038/nchembio.2254
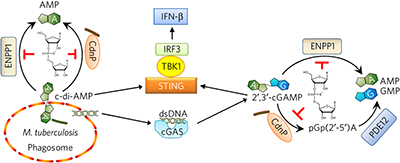
A mycobacterial phosphodiesterase, CdnP, hydrolyzes bacteria-derived 3′,5′-c-di-AMP as well as host-generated 2′,3′-cGAMP, which activates the host cytosolic surveillance pathway, to dampen host responses.
Chemical compounds
See also: News and Views by Li
|
|
|
 |
 |
 |
|
 |
 |
 |
|
 |
 |
 |
Orphan receptor ligand discovery by pickpocketing pharmacological neighbors pp235 - 242
Tony Ngo, Andrey V Ilatovskiy, Alastair G Stewart, James L J Coleman, Fiona M McRobb et al.
doi:10.1038/nchembio.2266
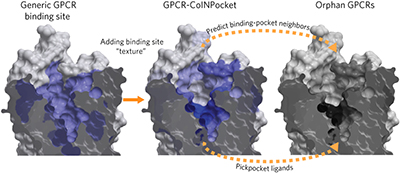
The 'CoINPocket' approach identifies pharmacological similarities between G protein-coupled receptors. On the basis of predicted pharmacological similarity to a few phylogenetically unrelated receptors, the approach identified surrogate ligands for the orphan receptor GPR37L1.
|
|
|
 |
 Top Top |
 |
 |
 |  |  |  |  |  | Natureevents is a fully searchable, multi-disciplinary database designed to maximise exposure for events organisers. The contents of the Natureevents Directory are now live. The digital version is available here.
Find the latest scientific conferences, courses, meetings and symposia on natureevents.com. For event advertising opportunities across the Nature Publishing Group portfolio please contact natureevents@nature.com |  |  |  |  |  |
|
 |


No comments:
Post a Comment